We use cookies to make your experience better. To comply with the new e-Privacy directive, we need to ask for your consent to set the cookies. Learn more.
BalanCD HEK293 Viral Feed
Boost viral vector production in HEK293 cells up to 10-fold.
The First Viral Feed Designed Specifically for HEK293 Cells
Gene therapy and viral vector-based vaccine manufacturers rely on basal media as the standard to grow their adeno-associated (AAV) viral vectors. The first-of-its-kind BalanCD HEK293 Viral Feed from FUJIFILM Irvine Scientific delivers a 3-fold to 10-fold increase in viral vector production over basal media alone to provide consistent performance, scalability, and speed time-to-market.


Scalable and adaptable viral vector performance
BalanCD HEK293 Viral Feed can be used with HEK293 suspension cultures from small- to large-scale for AAV production of multiple serotypes, as well as a range of agnostic basal growth media and transfection methods.
- Chemically defined BalanCD HEK293 Viral Feed, optimized to enhance viral vector production
- Supplement to HEK293 suspension cultures post-transfection and supports high cell densities
- A complete solution to achieve viral vector production goals for gene therapy applications
- Optimized for use with BalanCD HEK293 chemically defined medium and compatible with other basal growth media
Multiple-fold increases in viral titers
BalanCD HEK293 Viral Feed promotes vigorous VG/capsid yields and multiple-fold increases in titers to help manufacturers get more working volume for their applications.
AAV2 titer improvement upon feed
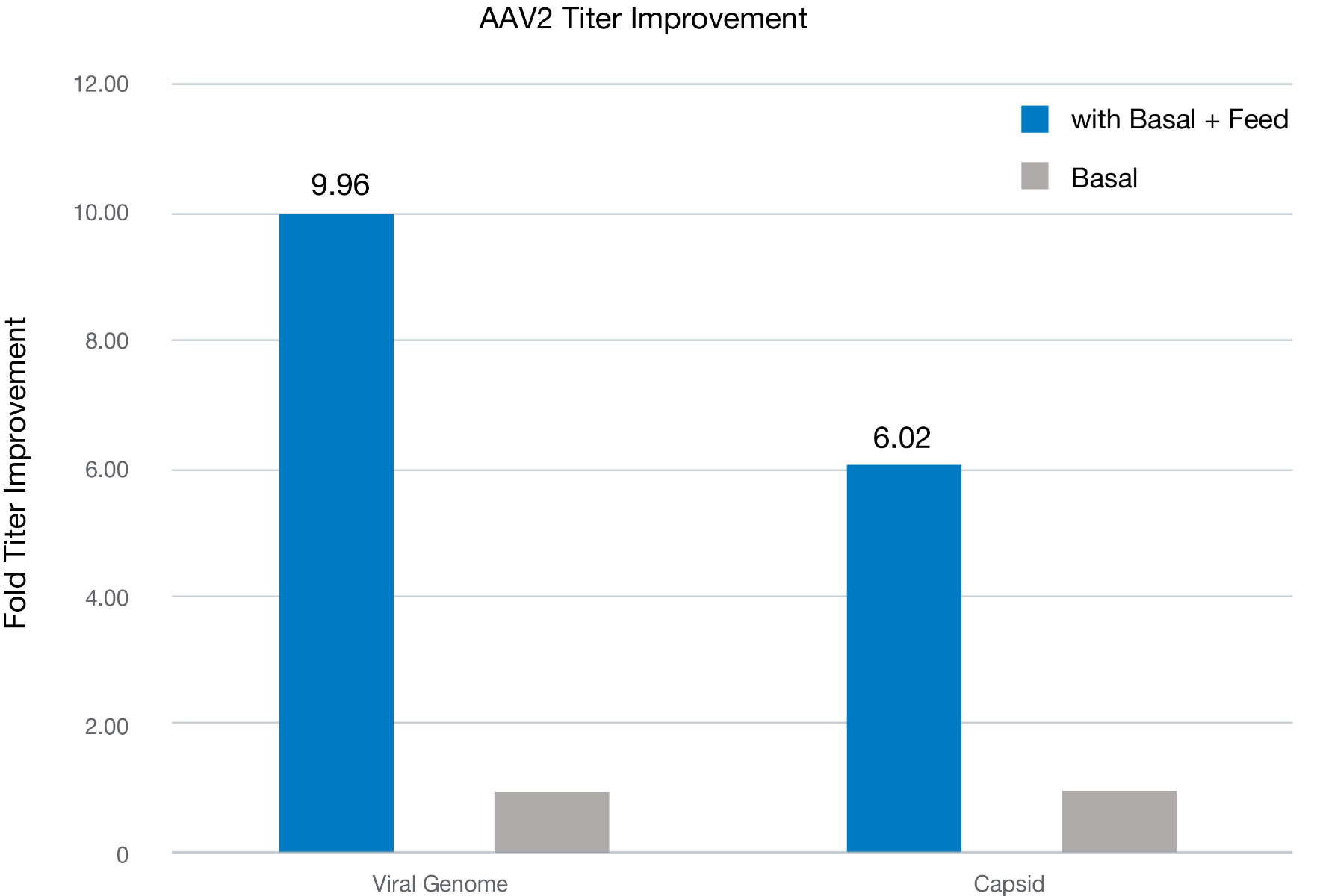

Figure 1. AAV2 viral genome titer and capsid titer were measured using cell extracts subjected to three cycles of freeze-thaw, followed by clarification by centrifugation and treatment with DNAse I and lysis buffer prior to qPCR (using AAVpro titration kit, Takara Bio) and ELISA (AAV2 titration ELISA, Progen). Blue and orange bars indicate the viral genome (VG) and capsid titer fold over the basal culture without a feed. The data indicates that the feed media enhancing the AAV2 production approximately 10-fold in VG and 6-fold for the capsid titer. As an overall result, viral packaging was determined to improve about 67%, from 13% in Basal to 21.81% in Basal+feed (data not shown).
BalanCD HEK293 Viral Feed increases the AAV titer in 10 liter scale-up
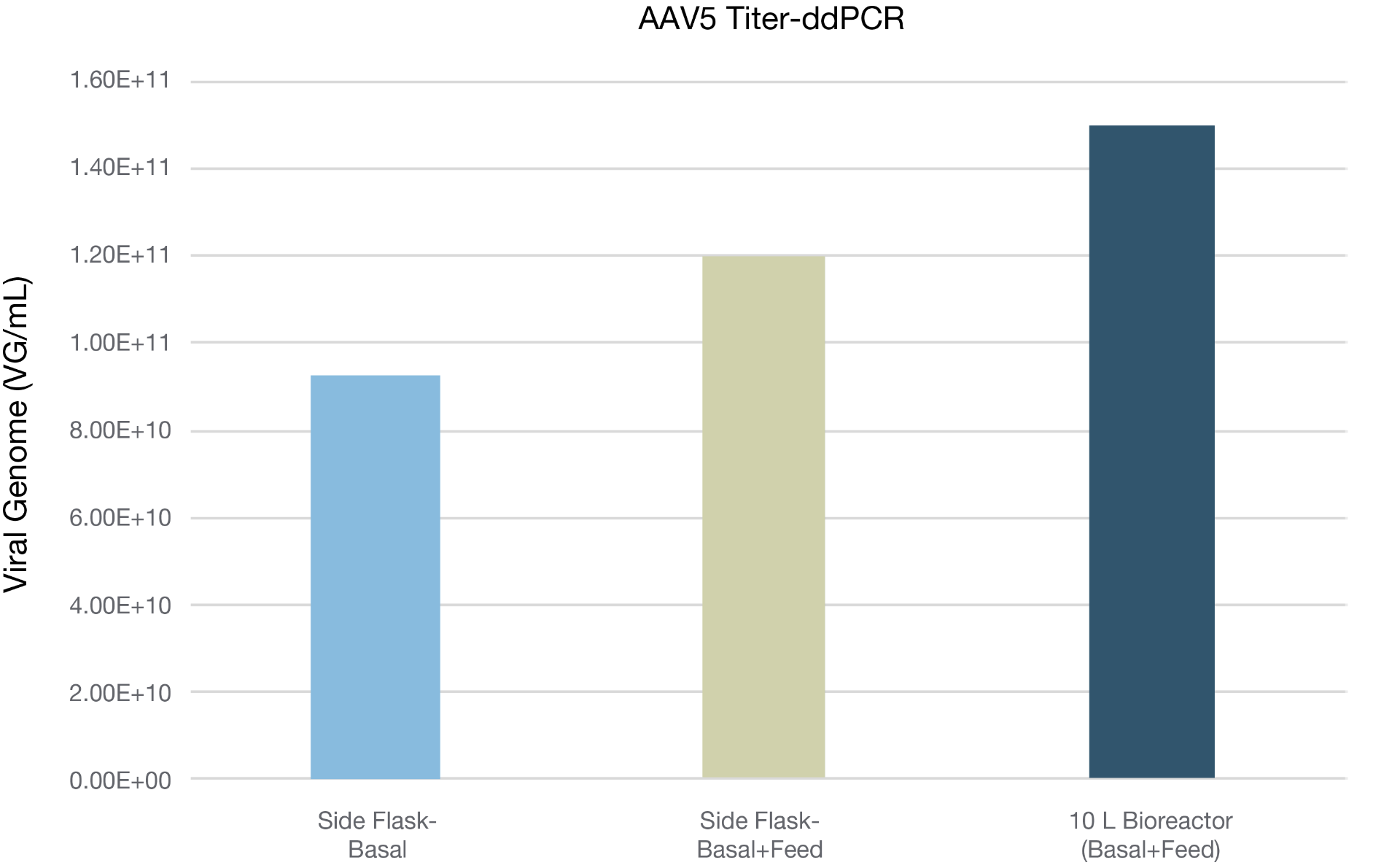

Figure 2. Verification of the BalanCD HEK293 Viral Feed performance on improving the production of AAV5 in 10L bioreactor scale. HEK293 suspension cells (cell line 2) were seeded at density of 0.8X10^6 cell/mL in 8.3 L culture volume in 10L bioreactor (XDR10 bioreactor). BalanCD HEK293 Viral Feed was added at 12% v/v at 24 hr post transfection. Titer was measured using ddPCR.
An uninterrupted supply of feed for HEK293 cell lines
BalanCD HEK293 Viral Feed is manufactured in a GMP facility using qualified raw materials sourced from a solid supply chain to ensure continuity of supply and lot-to-lot reliability for HEK293-specific applications. Our stringent oversight provides confirmation of formula, analysis, and assurance that BalanCD HEK293 Viral Feed meets the highest global and regional standards while fulfilling regulatory demands with each manufacturing lot file.
- Stringent raw materials control and sourcing program
- cGMP-compliant manufacturing
- COA, COO, TSE/BSE statements
- ISO13485, EN 13485:2016 certified
- Drug Master Files (DMF) supported
Contact us for other packaging sizes and powder format availability.