We use cookies to make your experience better. To comply with the new e-Privacy directive, we need to ask for your consent to set the cookies. Learn more.
FUJIFILM Irvine Scientific Introduces Sterile Express Media Service
The first manufacturing service of its kind delivers tested sterile, non-GMP media designed for feasibility testing before scale-up, to support development of cell and gene therapies
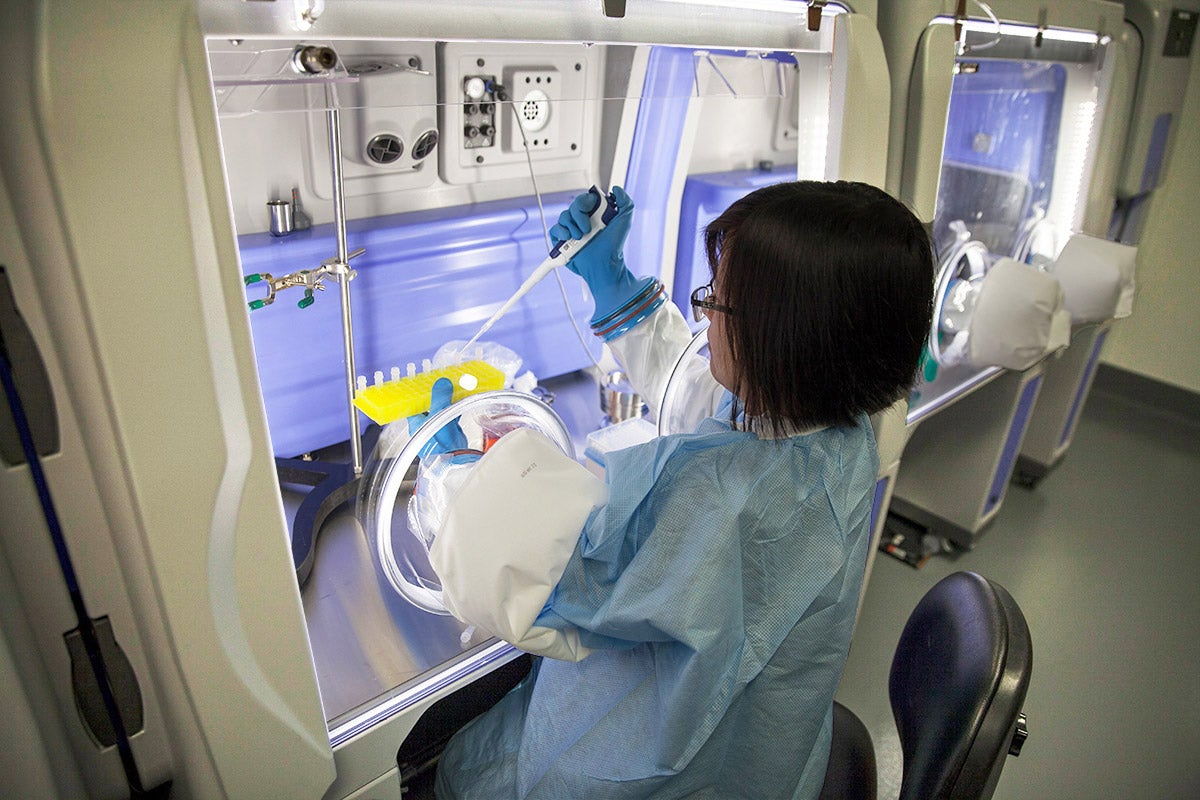
SANTA ANA, California, November 18, 2020: FUJIFILM Irvine Scientific, Inc., today announced the launch of Sterile Express Media Service (SEMS), for small-scale, rapid production of non-GMP media tested for sterility. With SEMS, customers can request up to 100L of media, to be rapidly manufactured in a Grade A/ISO 5 sterile environment, and have it shipped within 15 business days pending sterility confirmation.
Contamination in culture media can affect cell characteristics such as growth, metabolism, and morphology, contributing to unreliable or erroneous results. This can cause delays in the development and scale-up process. SEMS has been introduced to enable manufacturers to test the feasibility of formulations using sterile cell culture media before scale-up to more costly GMP manufacturing. Sterile media provide an extra measure of control and security for valuable cells and streamline the successful development of cell and gene therapies and biopharmaceutical drugs.
Unlike the sterile-filtered and sterile-tested media currently available in the market, SEMS utilizes isolator technology to provide an aseptic manufacturing environment. SEMS media remain in a Grade A/ISO 5 environment throughout the manufacturing process, reducing the risk of contamination for critical applications. The non-GMP liquid media formulations are manufactured using the same qualified raw materials used for GMP manufacturing and are sterility tested according to USP71 and ISO 13408 requirements.
"Until now, there has never been a rapid, non-GMP manufacturing service that delivers sterile cell culture media. Sterile Express Media Service is a first-of-its-kind offering that enables FUJIFILM Irvine Scientific customers to use sterile media for trials, which may also help reduce costs associated with developing therapies and biopharmaceutical drugs by reducing contamination risk during the testing phase. We are pleased to offer this innovation to our customers." – Erik Vaessen, Ph.D., Executive Director, Life Sciences, FUJIFILM Irvine Scientific.
SEMS is part of the FUJIFILM Irvine Scientific group of media services that also include Express Media Service (EMS) for small-scale, rapid production of non-GMP media and Media Development and Optimization Services (MDO) for the development of custom media.
For a high-resolution image please contact lorna.cuddon@zymecommunications.com
About Fujifilm
FUJIFILM Irvine Scientific, Inc.
FUJIFILM Irvine Scientific, Inc., is a worldwide leader in the innovation and manufacture of cell culture media, reagents, and medical devices for researchers and clinicians. The company provides unrivaled service and quality to scientists working in biopharmaceuticals, cell therapy, and regenerative medicine, assisted reproductive technology and cytogenetics, and industrial cell culture for the large-scale production of biotherapeutics and vaccines. FUJIFILM Irvine Scientific adheres to both ISO and FDA regulations and operates dual cGMP manufacturing facilities in California, USA, and Tokyo, Japan. The company’s consultative philosophy combined with expertise in cell culture and compliance provides customers with unique capabilities and support. For over 50 years, FUJIFILM Irvine Scientific has remained uniquely flexible and focused on media while becoming a strategic global leader in media products and services. FUJIFILM Irvine Scientific, Inc. is a subsidiary of FUJIFILM Holdings America Corporation reporting to FUJIFILM Holdings Corporation.
FUJIFILM Holdings
FUJIFILM Holdings Corporation, Tokyo, Japan, brings cutting edge solutions to a broad range of global industries by leveraging its depth of knowledge and fundamental technologies developed in its relentless pursuit of innovation. Its proprietary core technologies contribute to the various fields including healthcare, graphic systems, highly functional materials, optical devices, digital imaging and document products. These products and services are based on its extensive portfolio of chemical, mechanical, optical, electronic and imaging technologies. For the year ended March 31, 2020, the company had global revenues of $21 billion, at an exchange rate of 109 yen to the dollar. Fujifilm is committed to responsible environmental stewardship and good corporate citizenship. For more information, please visit www.fujifilmholdings.com.
Media Contacts
Lori Serles
FUJIFILM Irvine Scientific
Phone: (949) 261-7800 x145
Email: lori.serles@fujifilm.com
Lorna Cuddon
Zyme Communications
Phone: +44 (0)7811996942
Email: lorna.cuddon@zymecommunications.com

